人iPS干细胞分化来的肝细胞
|
| Cellartis enhanced hiPS-HEP v2 kits 提供了可持续供应的、成熟的人诱导多能干细胞(hiPS)来源的肝脏细胞。在超过14天的检测窗口期,hiPS-HEP细胞拥有功能性的药物代谢机制,使其成为体外药物研发、药物代谢、和相关毒性研究的理想选择;具备成熟、稳健的代谢特征,支持其作为代谢疾病模型。方便使用的试剂盒包括完全的培养基和组份,用于长期检测。与其他肝脏细胞模型相比,培养的Cellartis hiPS-HEP细胞存活时间更长,进而拥有更长的检测窗口用于慢性-毒性实验和作为代谢疾病模型研究。 |
| |
| ■ 产品特点 |
· Complete kit for thawing, plating, and culturing the provided cells
· Highly homogeneous population with consistent performance between batches and over time
· Cells are functional and stable for a 14-day assay window
· Cells show long-term, stable CYP450 activities
· Cells express key hepatocyte proteins, including α1AT, Alb, CK18, HNF4α, and E-cadherin
· Cells secrete physiologically relevant levels of albumin and urea
· Cells demonstrate functional insulin and glucose regulation
· Cells show phase I and II enzyme activities
· Cells demonstrate LDL uptake |
| |
| ■ 产品组成 |
· 1 vial of ~1.2 × 107 cells
· Hepatocyte thawing medium
· Hepatocyte coating
· Hepatocyte plating medium
· Hepatocyte washing medium
· Hepatocyte long-term maintenance medium |
| |
| ■ 产品应用 |
· Toxicity testing, including chronic toxicity assays
· Drug discovery and development
· Metabolic studies
· Metabolic disease modeling |
|
| |
■ 产品详情请点击: |
| |
 |
| |
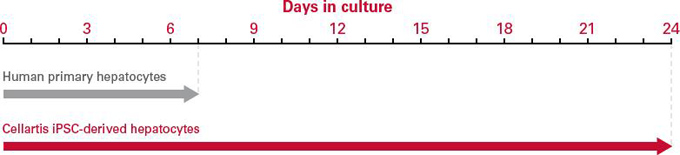
| Cellartis 人iPS细胞来源的肝脏细胞可以作为长期的、可靠的肝脏疾病模型。 |
| Cellartis human induced pluripotent stem (iPS) cell-derived hepatocytes are a long-lasting, reliable liver disease model. Ideal for long-term studies, these mature, functional, pure hepatocytes allow you to generate consistent results with low batch-to-batch variability. Cellartis iPSC-derived hepatocytes can be used for extended culture significantly longer than human primary hepatocytes—allowing you to get more data from your chronic toxicity studies. |
| |
 |
| |
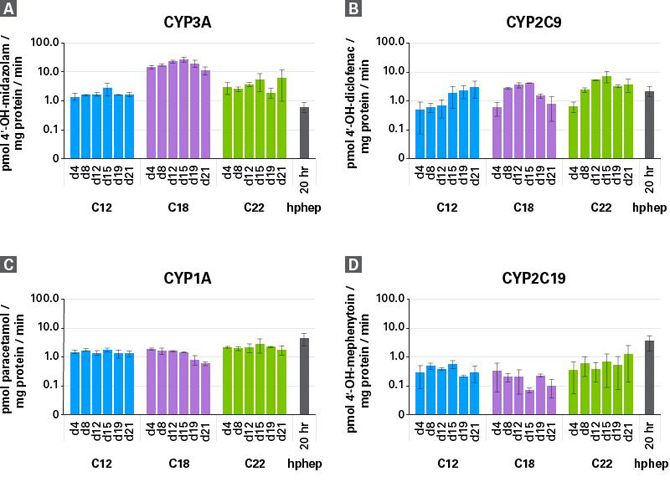
| Cellartis enhanced hiPS-HEP cells可以稳定表达CYP450 活性超过21天。 |
| CYP450 activity is stable in enhanced hiPS-HEP cells over 21 days. LC/MS was used to analyze CYP450 activity in cultured enhanced hiPS-HEP cells previously derived from the hiPS cell lines ChiPSC12, ChiPSC18, and ChiPSC22 (abbreviated as C12, C18, and C22). CYP3A (Panel A), CYP2C9 (Panel B), CYP1A (Panel C), and CYP2C19 (Panel D) activities in enhanced hiPS-HEP cells are stable over an extended culture time. Cryopreserved human primary hepatocytes (hphep), which are functional in culture for a significantly shorter time than enhanced hiPS-HEP cells, were thawed and cultured for 20 hours, and their data are shown as the black bar in each panel. |
| |
 |
| |
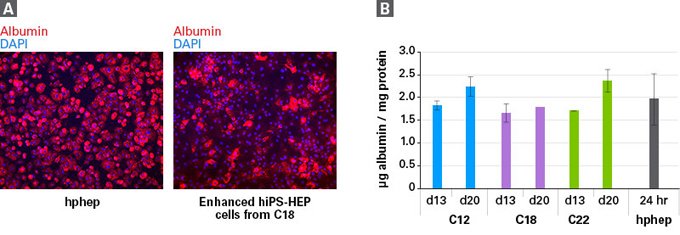
| Cellartis enhanced hiPS-HEP细胞在20天的培养中表达白蛋白(Albumin)。 |
| Albumin is present in enhanced hiPS-HEP cells through 20 days in culture. Panel A. Representative images of enhanced hiPS-HEP cells from human iPS cell line ChiPSC18 (C18) taken 12 days after thawing (right), compared to cryopreserved human primary hepatocytes (hphep) taken 24 hr after thawing (left). Cells were stained for albumin and DAPI. Panel B. Albumin secretion as measured by ELISA; n=2 for enhanced hiPS-HEP cells, with the exception of C18 at 20 days (n=1), and n=3 donors for hphep. |
| |
| (以上图片均来源于Takara Bio USA, Inc.) |
| |
| 参考文献: |
1. Heins et al. Stem Cells 2004; 22: 367-376.United States National Stem Cell Bank; http://www.nationalstemcellbank.org.
2. Mantel N et al. Potential markers of attenuation of YF virus after infection of stem cell-derived human hepatocytes with wild-type Asibi or live-attenuated YF17D virus.Supplement to The American Journal of tropical Medicine and Hygiene, Volume 83, November 2010, Number 5, abstract 12.
3. Yildirimman R et al. Human embryonic stem cell derived hepatocyte-like cells as a tool for in vitro hazard assessment of chemical carcinogenicity. Toxicol. Sci. 2011 Dec; 124(2): 278-90.
4. Ulvestad M et al. Drug metabolizing enzyme and transporter protein profi les of hepatocytes derived from human embryonic and induced pluripotent stem cells. Biochem Pharmacol. 2013 Sep 1; 86(5):691-702. |
| |
| |